 The blue lines on the graph represent the pressure lines of the material. In this specific graph, there are three sets of colored lines going across the graph. The inverse is also true as known physical value properties allow for the determination of enthalpy or entropy. The graph allows these property determinations from known entropy and enthalpy values. The lines on the chart provide all possible ranges of different values such as pressure, temperature, or even quality. Graphing enthalpy vs entropy allows for the determination of some key properties of the subject material. It calculates as the combination of the internal energy( E) with pressure( P) and volume( V). This classifies the property as a non-physical property. Practically, it measures a change in total internal energy. EnthalpyĮnthalpy represents the total energy within a substance, however, it is impossible to directly measure. Understanding enthalpy and entropy clarify what these differences mean and when to use each measurement. Where enthalpy is a measurement of energy potential, entropy measures the randomness of energy with relation to heat. 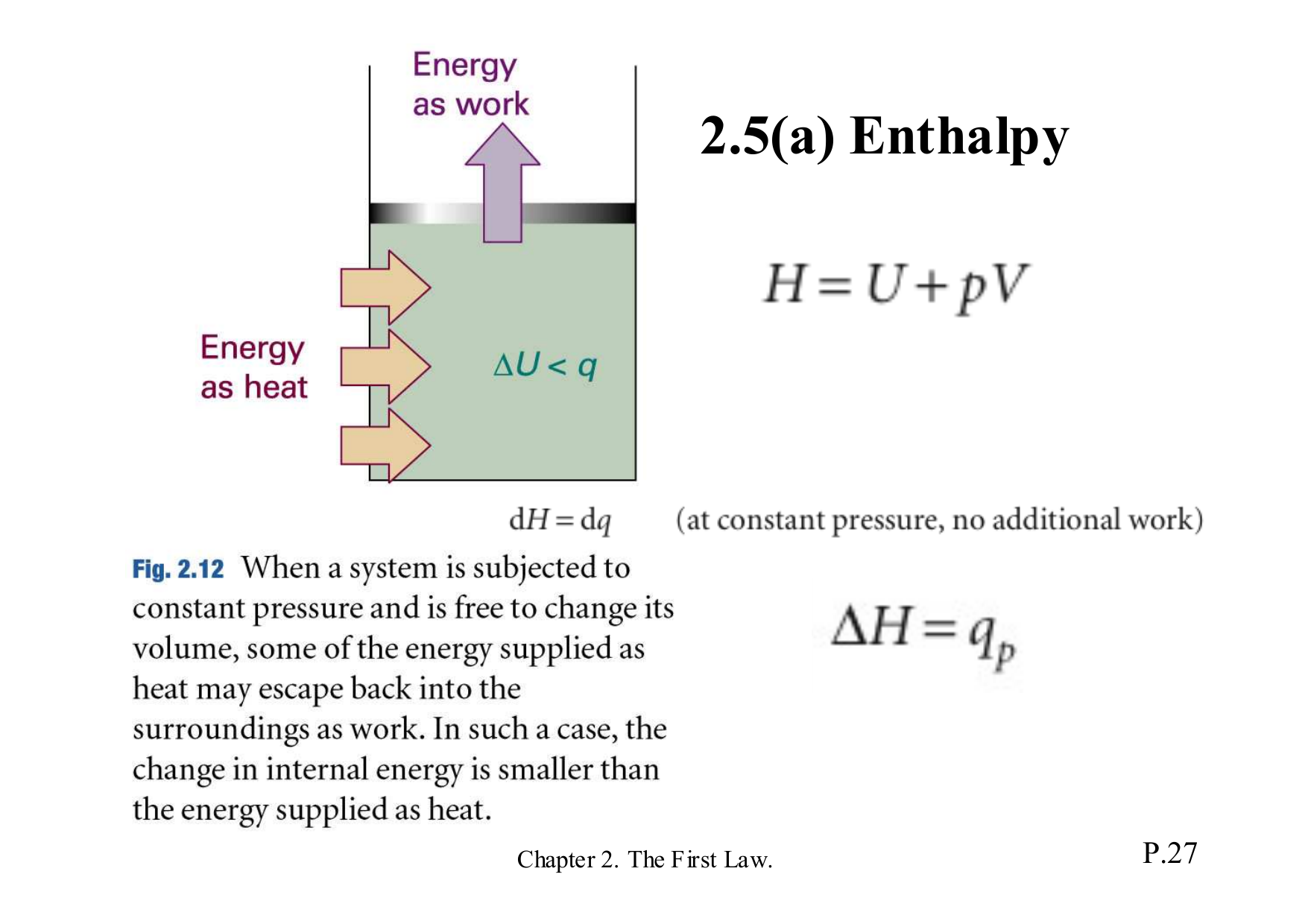
COURTESY: THOUGHT CO Differences of Enthalpy and Entropy In this article, you will learn the differences between enthalpy and entropy, the H-S Chart, and applications of these properties in the real world. Entropy measures the randomness in the material, also understood as the unavailability of energy, within the said compound. Enthalpy provides the total amount of internal energy held within a substance. Enthalpy vs entropy -what’s the difference? All materials contain both of these thermodynamic properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
